Peripheral Magnetic Stimulation (PMS) Therapy
Non-Invasive, FDA-Cleared Relief for Chronic Nerve and Pain Conditions
Peripheral magnetic stimulation (also called magnetic peripheral nerve stimulation, mPNS or PMS) is a non‑invasive, drug‑free treatment that uses pulsed magnetic fields to calm irritated nerves, interrupt pain signals, and help restore normal function. It is FDA‑cleared for the stimulation of peripheral nerves to relieve chronic intractable, post‑traumatic, and post‑surgical pain—and for painful diabetic neuropathy of the lower extremities in adults.
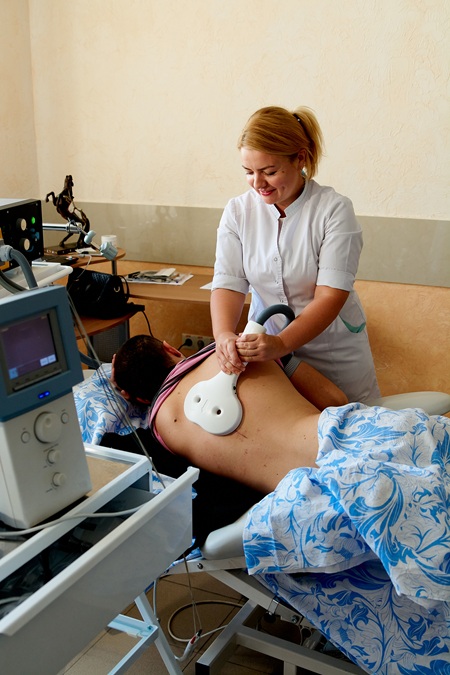
How Peripheral Magnetic Stimulation (PMS) Works
During treatment, a specialized coil is placed on the skin over the area of pain or along the path of a peripheral nerve. Brief, rapidly changing magnetic pulses pass through soft tissue and induce tiny electrical currents in the underlying nerve fibers without needles, surgery, or medications.
These currents modulate how nerves fire, helping to “recondition” pain pathways and reduce abnormal pain signaling to the brain (a process called neuromodulation). Research shows that magnetic peripheral nerve stimulation can reduce neuropathic pain intensity by around 50% on average after a treatment series, with responder rates (patients achieving at least 50% pain reduction) around 70% or higher in some trials. Sessions are typically short—often in the 15 – 30 minute range—with minimal side effects such as mild tingling or muscle twitching during treatment and no required downtime.
What conditions it can treat
Magnetic peripheral stimulation is used for a variety of chronic pain and nerve‑related conditions, especially when medications, physical therapy, or injections have not provided enough relief.
Common indications include:
- Chronic intractable pain
- Post‑surgical and post‑traumatic nerve pain
- Painful diabetic neuropathy of the lower extremities
- Complex regional pain syndrome (CRPS)
- Post‑amputation (phantom limb) pain
- Occipital neuralgia and certain headache‑related nerve pains
- Osteoarthritis‑related joint pain and some forms of chronic low back pain
In a multi‑center randomized trial of patients with painful diabetic neuropathy, 71% of those receiving mPNS plus standard care achieved at least 50% pain reduction at 90 days, versus 13% with standard care alone. Long‑term follow‑up showed that by one year, 94% of patients who continued therapy were responders, and over 60% experienced greater than 80% pain reduction. Other clinical studies on peripheral magnetic stimulation report pain score reductions ranging from about 60% up to complete relief in some patients, with no serious therapy‑related adverse events.
Quick benefits snapshot
- Non‑invasive, no incisions, implants, or injections.
- Drug‑free option that may help reduce reliance on pain medications.
- Short treatments (often around 13.5 minutes), with relief that can last weeks to months in many patients.
- FDA‑cleared specifically for chronic intractable, post‑traumatic, post‑surgical pain and painful diabetic neuropathy in adults 18+.
Is peripheral magnetic stimulation FDA‑cleared?
Yes. Magnetic peripheral nerve stimulation systems have received FDA clearance for stimulating peripheral nerves to relieve:
- Chronic intractable pain
- Post‑traumatic and post‑surgical pain
- Painful diabetic neuropathy of the lower extremities (in adults 18 and older)
FDA clearance means the device and treatment are recognized as safe and effective for these specific indications when used as directed. This non‑opioid, non‑surgical option is part of a broader effort to expand evidence‑based, safer pain management strategies for the millions of Americans living with chronic pain.
FAQs About Peripheral Magnetic Stimulation (PMS)
Does treatment hurt?
Most patients describe treatment as a tapping or gentle pulsing sensation under the coil, with occasional muscle twitching, but not as painful.
How many sessions will I need?
Protocols vary, but many patients start with a short series over several weeks (for example, multiple sessions over 1–2 months), then transition to maintenance sessions every 6–8 weeks if needed.
What results can I expect?
Individual results vary, but clinical trials show that about 7 out of 10 patients can achieve at least 50% reduction in neuropathic pain after a full treatment course, with some reporting greater than 80% improvement and better daily function. Some patients notice early relief after the first few sessions; for others, improvement builds gradually over several weeks.
Is it safe?
Yes, when delivered with FDA‑cleared systems under appropriate clinical supervision, peripheral magnetic stimulation has a strong safety profile, with low rates of mild, transient side effects and no surgery or anesthesia required.
Who is not a candidate?
People with certain implanted electronic devices (such as some pacemakers or neurostimulators), metal near the treatment site, or specific medical conditions may not be candidates and should be carefully screened by a provider.
Why Choose PMS?
If chronic nerve‑related pain is limiting your life, peripheral magnetic stimulation may be a safe, FDA‑cleared option to finally get lasting relief without surgery or long‑term medications. To find out whether you are a good candidate, schedule a consultation with a qualified provider, bring a list of your current medications and medical history, and ask how magnetic peripheral nerve stimulation could fit into a personalized pain‑management plan tailored to your goals.
Final Thoughts
Bursa injections are a powerful, minimally invasive option for relieving bursitis pain and restoring function. With expert guidance, advanced imaging, and a focus on rehabilitation, patients can expect rapid recovery and lasting improvements in mobility and quality of life.
Let’s realign your spine and restore your quality of life. Book a personalized evaluation today.
Where Does It Hurt? We Can Help.